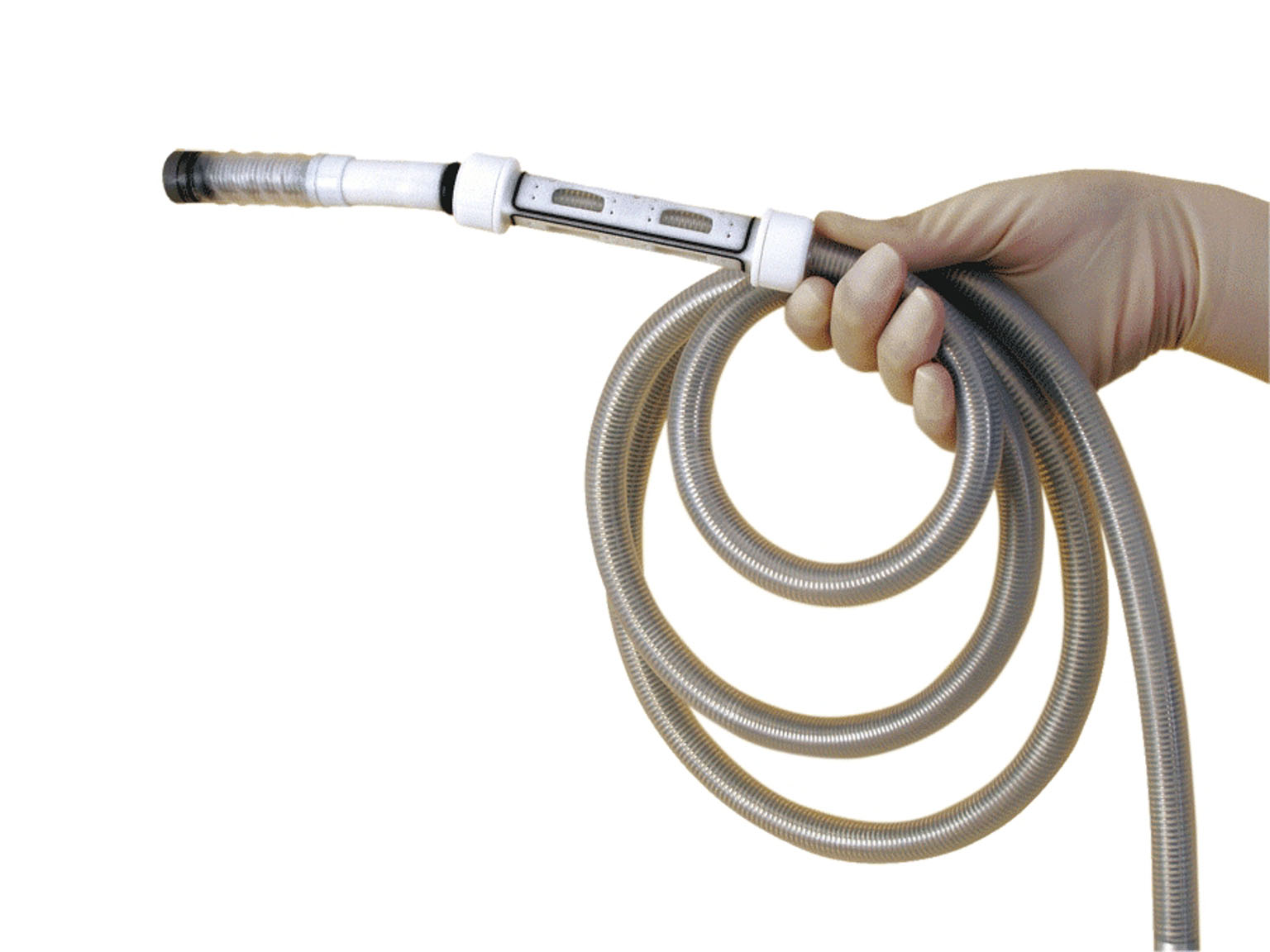
Invendoscope
On a February morning at New York Presbyterian’s gastroenterology suite, John Cifarelli rests on his side, knees up, buttocks barely hidden by a pair of shorts with an easy-access panel. Suit-clad executives surround him. Whatever happens next would determine whether Cifarelli’s faith in his employer was well founded.
Having worked in the medical device industry for the better part of 25 years, Cifarelli knows how difficult it is to break free of the novelty news stories about advancing technology. In many cases, innovators get a day’s worth of media mentions before finding out that their latest gadget doesn’t stand up to clinical or Food and Drug Administration (FDA) scrutiny.
It was impossible to say whether things would be any different with Invendo, the company Cifarelli joined in 2006. Well-versed in the gastroenterology field, Cifarelli was well aware of the many devices that had tried (and failed) to improve on the dreaded colonoscopy, an invasive but oft-prescribed screening procedure involving a camera, a six-foot-long tube, and one’s anus. Invendo had promised to make the exam more comfortable, more sanitary, and sedation-free.
Cifarelli was charged with promoting the device in the United States for the German-based company. He knew he would have to speak from experience if he were to have any credibility with hospitals or providers. This is why he found himself on a gurney and preparing to undergo a procedure that more than 20 million adults have refused out of fear or anxiety. And why he planned to do it completely sober, in front of eyewitnesses.
“I was trying to be a company guy,” he says of the audience. “But everyone and his brother were in the room.”
Cifarelli took a deep breath, stared at the bare wall opposite him, and braced himself.
Made in Japan
Hiromi Shinya, a Japanese gastroenterologist and surgeon, completed his residency in the late 1960s at Beth Israel Medical Center in New York. For reasons unfathomable, he and a colleague, William Wolff, MD, decided to poke an esophageal scope up rectums and see what was what in there. By 1969, Olympus had modified the scopes to better navigate the twists of the intestine, and Shinya could get to the cecum — the end of the large bowel, which looks like the Sarlaac pit from Star Wars — most of the time. The colonoscopy was born.
Performed early enough, the invasive exam can save lives, as it can include a procedure to snip sprouting polyps before they have an opportunity to ravage your body: colon cancer is the second leading cause of cancer deaths worldwide. Performed regularly enough, colonoscopies also represent a profitable and pervasive part of a gastroenterologist’s practice.
Though less uncomfortable tests have floated around in the margins for years, a definitive answer resides only with the “gold standard” and its accompanying inconveniences: self-induced diarrhea, to clean out the colon, and “twilight” sedation, during which you may or may not have a vague feeling of being violated. The former keeps you housebound for an evening; the latter means missing an entire day of work.
Aside from advancements in cameras and flexibility, the colonoscopy has remained largely the same since Shinya first went spelunking in the ’60s. “There’s been no dramatic change in 40 years,” Cifarelli says. “I can’t think of any other medical instrument you can say that about.”
The most promising alternative to optical colonoscopy was the emergence of CT colonography, or radiological imaging of the colon. Studies have shown a detection level comparable with conventional examination. President Obama opted for the CT scan in 2010, causing a mild stir: agitated gastroenterologists, sensing radiologists encroaching on their territory, pointed out that a growth spotted during a scan can’t be removed, while it can with a traditional colonoscopy.
With CT a viable but limiting alternative — its detection rate is highly dependent on the skill of the radiologist reading the data — thoughts turned toward making a conventional exam more tolerable. Several companies tried to develop scopes that reduced pressure on the colon wall, reducing pain and the need for sedation. One prospect, the Aer-O-Scope, is a camera bookended by two balloons. One “seals the anus” to provide a pressurized atmosphere to propel the other, which looks like a puffer fish; another tried to mimic the subtle locomotion of the inchworm, crawling the bowel like a parasite.
The problem, according to Cifarelli, is that some of these devices were too divorced from the accepted method to ever gain traction. “The Aer-O-Scope was self-advancing,” he says, “but it wasn’t physician-controlled. That’s going to require a lengthier FDA process.”
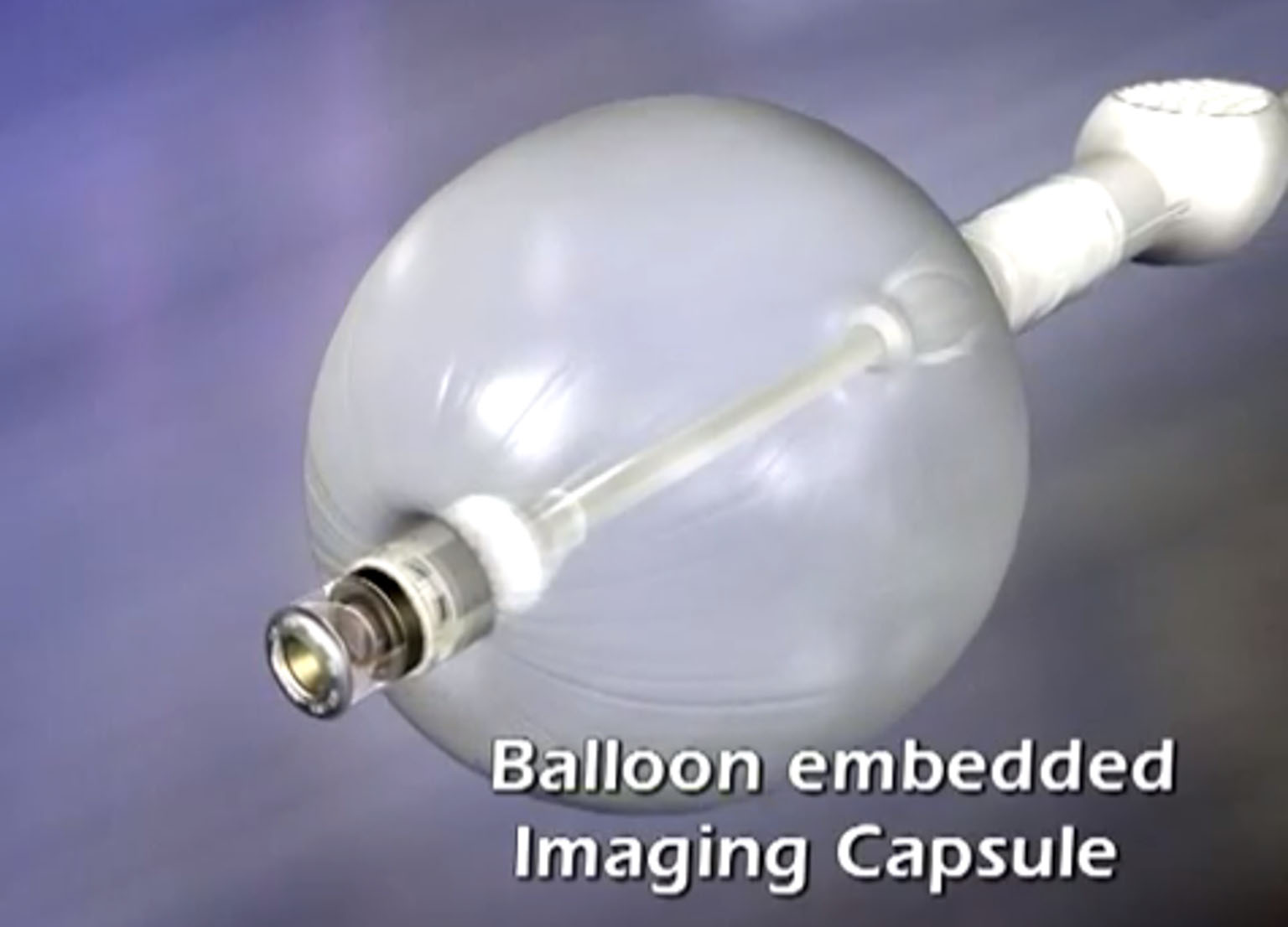
The Aer-O-Scope
Invendo avoided this by reinventing the classic scope. Researchers recognized that most of the discomfort from the exam came from “looping,” or the tube getting bunched up in the intestine as it tries to navigate four sharp turns in the colon. (Imagine trying to push a semi-cooked spaghetti strand through a maze.) What if, the company posited, their scope simply extended itself using a motor without the physician pushing and pulling on it like a plumber snaking a drain?
The Invendoscope is best imagined as resembling E.T.’s finger. The one-time-use tube expands rather than pushes through the colon, reducing looping and allowing for a more comfortable exam. Physicians remain in control by using a remote. After showing efficacy comparable to colonoscopy in German trials, the device was granted FDA approval in 2012.
The Invendoscope is currently housed at two major hospitals: New York Presbyterian and New York University-Langone, where Mark Pochapin, an MD, is championing its use. (Pochapin may be best known for scoping Katie Couric and Harry Smith during two particularly disturbing Today show segments intended to spread the gospel of colonoscopy, both completely at ease with having a doctor wash away bits of their fecal residue on national television.)
It’s Invendo’s hope that the prestige of these hospitals will pique patient curiosity and lead to more installations around the country. “The biggest challenge for us is awareness,” Cifarelli says. “Patients are not aware other modalities exist.”
But general population acceptance must be preceded by clinician acceptance. Cifarelli knew that the industry was dominated by Olympus, with close to 80 percent of the colonoscope market share. Worse, physicians are wary of anything that disrupts the status quo.
“This test has been great to them, and they don’t want to see change,” Cifarelli says. “It also needs to make sense to a hospital administration and to patients.” Many flee the exam room knowing they’ll be put to sleep. Cifarelli’s job depends on whether they’re willing to do it awake.
Awakenings
In German studies, patients who found the Invendoscope untenable asked for and were granted the standard intravenous drugs used for such exams. Properly titrated, Versed or Propofol provide sufficient relief from the diagnostic roto-rooting, something a painkiller cannot provide.
“Some people would just as soon be asleep, independent of the pain,” says Michael B. Wallace, a professor of medicine at Mayo Clinic in Florida. “It’s partly the embarrassment. There’s a broader tendency here in the States to want to be sedated for uncomfortable things.”
Wallace notes he had his own conventional and gimmick-free colonoscopy done without drugs, but varying operator skill levels and colon shapes — women tend to have particularly “twisty” bowels — make it hard to predict who will find a sober exam tolerable and who will need some kind of post-event psychotherapy
Gicare Pharma believes this will soon change. A Phase II clinical trial spearheaded by the Mayo Clinic is currently underway for the company’s GIC-1001, which sounds like a MacGuffin for a Hitchcock film but is instead a proprietary blend of local analgesics that render the colon (and only the colon) incapable of feeling pain.
Volunteers who take GIC-1001 typically ingest it for three days leading up to the exam, and once more on the day of. The drug saturates opiate receptors in the intestinal tract, giving the colon a localized numbing effect. Gicare Pharma’s expectation is that it will relieve all associated pain and discomfort from the exam, including the sense of bloating provided by air pushed in to inflate the colon, while only costing half of that of conventional sedation.
Maxime Ranger, the CEO of Gicare Pharma, underwent a colonoscopy in Canada using only Modulon, one of the drugs in GIC-1001’s more potent cocktail. “I was quite surprised,” he says. “It took only six minutes to reach the cecum. The drug also has an anti-spasmodic effect, which reduces movement. The patient has no recovery time and can drive themselves home.”
If GIC-1001 passes its second clinical study without anyone sprouting horns, another trial will get underway in 2015, using a targeted dosage amount titrated in phase II. The best-case scenario, Ranger says, is to have a potentially uneventful conscious exam with the FDA’s blessing by 2017. It could either complement Invendo’s efforts or steamroll them.
Tough to swallow
Canada, along with frontrunners Germany and Japan, seems to have a preoccupation with bowel visualization. It was one of the first countries to embrace Given Imaging’s PillCam COLON 2, which does away with the scope entirely and enters into the territory of 1960s sci-fi spinner racks. Patients who wish to have their innards investigated swallow a pill the size of a shotgun shell. Tiny cameras on each end take thousands of pictures as the device tumbles, directionless, through the digestive tract.
Matthew Schroyer, a technology blogger who has periodic flares of Crohn’s disease, discovered the PillCam when his physician was unable to adequately visualize a portion of his colon that was narrowed due to the inflammatory disease. “I was familiar with research being done on the feasibility of it, with cameras getting smaller,” he says. “But I didn’t know it had come all the way to the consumer end. It sounded cool to me.”
After a nurse affixed a recording device to his chest, Schroyer swallowed the PillCam with a cup of water, its tiny LED lights blinking a preemptive distress signal. Eight hours later, he was back in the office to return the hard drive and collect a warning. “If you don’t see this come out by tomorrow,” the nurse said, “then we’ll have to go in after it.”
This ominous hypothetical proved to be unnecessary. Schroyer passed the capsule camera, cleaned it off, and like any good techie, promptly tried hacking it. “It was dead,” he says disappointedly. “You apparently can’t get it to work again after the battery goes. Probably a safety feature.”
The next week, Schroyer and his physician reviewed the images. There was pus and redness where there should be no pus and redness. The doctor plotted a course of treatment and put his Crohn’s in remission. Schroyer keeps the capsule on his desk.
Aside from reviewing images, the PillCam demands little from a gastroenterologist: a nurse can easily administer the capsule and tighten the straps on the hard drive. It’s that kind of autonomy that Cifarelli believes will help drive the Invendo and other devices in the coming years.
“A nurse could operate the Invendo,” he says. “And if demand peaked, if all the people who were referred for the exam got one, there wouldn’t be enough gastroenterologists to meet the demand.”

PillCam
Future inspections
The PillCam, unlike the Invendo, is diagnostic only. It cannot intervene. Worse, it cannot move in for closer inspections, tumbling down the GI tract and snapping pictures at rapid intervals with no guiding mind. It is an ignorant device, incapable of being controlled, even by a trained specialist.
“Even the best bowel prep leaves a little bit of mucus or liquid stool that obscures the colon wall,” says Wallace. “With a scope, we can wash it out. With a passive system, you can’t rinse, stop, go back and look.”
The real potential alternative to colonoscopy remains uncertain. Wallace says that precious little data exists on Invendo’s claims of a more comfortable exam. And while the PillCam just received FDA approval and should begin popping up in specialist offices soon, those with swallowing difficulties might panic.
Wallace instead has placed his hopes on a DNA stool test currently under FDA evaluation that looks for specific cells shed by tumors. “It’s hard to know what the FDA’s decision will be,” he says. “The test is not as accurate as colonoscopy, but it’s totally non-invasive. We should see a decision within the next six months.”
It seems likely that the next several years will see a substantial change to the landscape of colonic inspection: screening rates, according to the Centers for Disease Control, are still too low, and the potential to make money is too high. The proposed practice of “fecal tagging,” which sounds like a horrible job at the Gap, might serve to eliminate bowel prep for CT scans; a hydro-colonic device at NYU might, in one flush, do away with hours of unpleasant rectal weeping.
Young minds at Vanderbilt University are already experimenting with the Magnetic Air Capsule (MAC), a tiny robot that is controlled by magnets and resembles a Swiss Army knife. At some point, its researchers might graduate from scoping pig intestines and move to human trials. A protein test developed by Applied Proteomics and said to be remarkably accurate in detecting tumor markers in blood may hit the market as soon as this year.
Though it’s obvious Invendo and others want to win this gastrointestinal arms race, “There is no one absolute right answer,” Wallace says. “If you really don’t want a colonoscopy, you do have other choices.”
The end result
In the gastroenterology suite, Cifarelli waits for the pain. The executives watch as the device is inserted. Cifarelli felt some air pressure, “like a gas bubble,” but it quickly passed.
In no acute distress, he reached for his phone and emailed a curious reporter — me. “I am in my procedure,” his note read. “We reached the cecum. I am emailing you as the procedure is on. I am fine.”
“It was better than a teeth cleaning,” he says. Noting that the physician took his time owing to the audience, he did not experience anything painful. “It felt less intrusive than a digital rectal exam.”
There is no telling if the Invendo or the PillCam or a designer drug will be the next alternative to what seems like an outdated procedure. But Cifarelli hopes physicians will be more vocal about the alternatives, particularly when they can literally save the day for patients who often spend hours shaking off the cotton-headed feeling of sedation.
“I hopped off the table,” Cifarelli says, “got dressed, and had lunch.”
Jake Rossen has written for a variety of publications including the New York Times, the Village Voice, ESPN the Magazine, Mental Floss, and MIT Technology Review.
